The unmistakable hiss of an aerosol can echoing across a crowded beach is the soundtrack of July. It smells like coconut, salt spray, and the promise of a sunburn-free afternoon. You shake the icy metal cylinder, feeling the liquid slosh inside, then press the nozzle and sweep a fine, cooling mist across your shoulders. It feels like an act of care, a quick chore checked off before returning to the water. For years, we have embraced this ritual without a second thought, trading the messy, sticky hands of traditional lotions for a seamless, continuous spray that vanishes into the skin almost instantly.
But beneath that breezy application, a silent reaction is brewing. We have been taught to trust the convenience of the spray, assuming the liquid inside remains pristine until it touches the air. The harsh reality of modern cosmetic chemistry tells a different story entirely, one that is currently sending shockwaves through the sun care industry.
The metal canister you tossed into your canvas tote is not a simple container. Sitting in a hot car or baking on a pool deck, the tightly sealed, pressurized environment forces chemical propellants to mingle intimately with delicate UV filters. In a sterile laboratory kept at exactly sixty-eight degrees, these components behave. But out in the wild, under the baking summer sun, the canister transforms. What emerges from the nozzle is no longer the gentle, protective lotion you originally purchased.
Instead of wrapping you in a protective blanket, these compromised aerosol formulas are quietly degrading into harsh, reactive irritants. The recent wave of recalls and reports of severe skin reactions is not a fluke of a bad batch. It is the natural consequence of putting highly reactive sun care into a volatile environment.
The Pressure Cooker On Your Towel
Think of the aerosol can not as a bottle, but as a miniature pressure cooker. When you mix delicate chemical compounds meant to absorb solar radiation with liquid gases designed to propel them out of a tiny hole, stability becomes a fragile illusion.
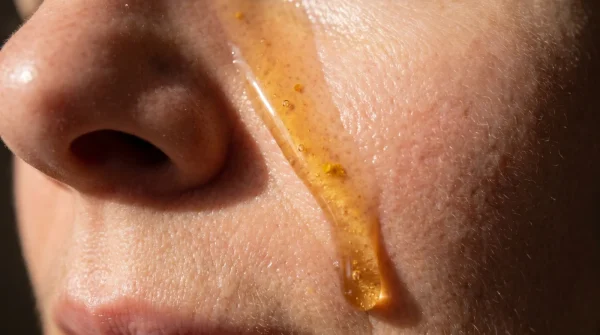
You might assume sunscreen is a static shield, but it acts more like a living battery. When subjected to the ninety-degree heat of a beach bag, those propellants begin to slowly break down the protective filters. The ingredients meant to diffuse ultraviolet light instead turn inward, oxidizing and forming new, acidic compounds. You are inadvertently spraying a microscopic chemical burn directly onto your delicate skin.
Aris Thorne, a forty-eight-year-old cosmetic chemist based out of a quiet lab in coastal New Jersey, spent over a decade stabilizing these exact mists. He recalls pulling older spray cans from heat-testing incubators and finding that the pH levels had plummeted, turning mild lotions into biting acids. Aris often tells his colleagues that asking complex UV filters to survive inside a pressurized gas chamber is like asking a delicate pastry to bake perfectly inside a washing machine on the spin cycle. He noticed that the combination of butane propellants and organic chemical filters creates an incredibly fragile emulsion. The degradation is not an accident; it is just basic thermodynamics playing out in your beach tote. When you understand the science, the recent product recalls stop looking like corporate mistakes and start looking like an inevitable breaking point.
How the Fallout Affects Different Lifestyles
The way this chemical degradation manifests on your skin depends heavily on how you spend your time outdoors. The reaction is not uniform, and understanding your specific vulnerability is the first step toward making safer choices for your body.
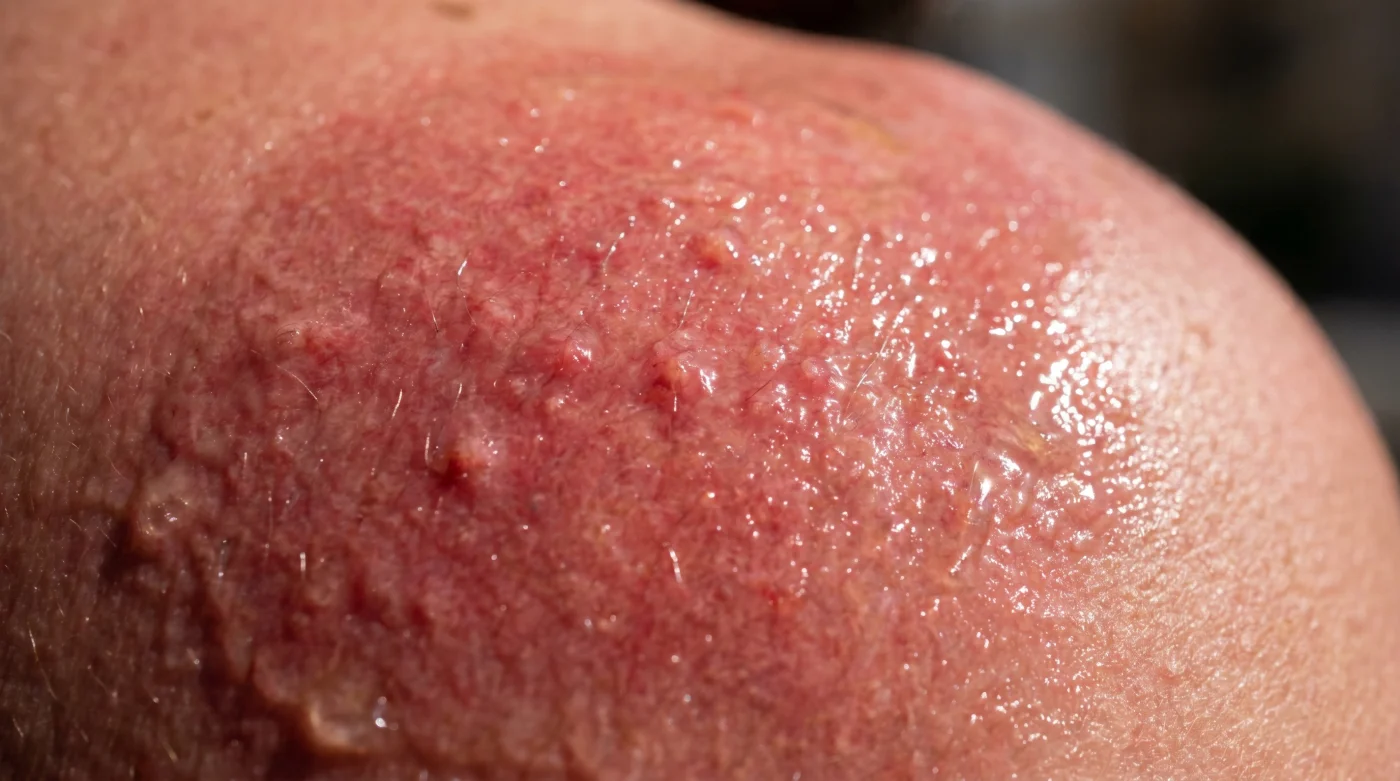
For the purist and sensitive skin types, these degraded mists act as a match in a dry forest. If you already suffer from rosacea or an easily disrupted skin barrier, the newly formed acids compromise your lipid defenses almost instantly. You might feel a tightening sensation followed by an intense, prickling heat. It leaves you with patchy, stinging red marks that you might incorrectly blame on the sun itself, entirely unaware that the mist you applied to prevent the burn is exactly what caused it.
For the marathon runner and hiker, heavy sweating changes the dynamic. When you perspire, salt mixes with unstable chemicals to create a highly frictional brine. This heavy, acidic sweat traps the irritating compounds against your pores, often resulting in deep, painful micro-blisters along the back of your neck or behind your knees as you rack up miles.
- Michelle Obama spring fashion choices disrupt traditional power suiting rules
- Baking soda scrubs permanently alter your facial acid mantle within days
- Retinol serums destroy overnight repair cycles if applied over heavy moisturizers
- Liquid collagen supplements require vitamin C to actually rebuild thinning hair
- Jade rollers spread microscopic bacteria unless aggressively sanitized in boiling water
- Coconut oil silently destroys your natural moisture barrier with daily application
- Green tea bags drastically reduce under-eye puffiness better than luxury creams
- Micellar water requires immediate rinsing to prevent severe microscopic pore congestion
- Hyaluronic acid actually accelerates deep wrinkle formation without totally damp skin
- Germany bans common sunscreen ingredients hiding in American luxury cosmetic lines
For the casual weekend lounger, you might not notice the chemical burn immediately. Instead, as you lazily reapply layer after layer of the compromised mist while reading by the pool or sipping a cold drink, you are slowly accumulating a toxic trace on your shoulders. The irritation builds quietly, mimicking a mild windburn. It feels tight and slightly warm, easily ignored until you pack up your towel, head inside, and step into a hot shower. Only then do you feel the unmistakable, stinging consequence of degraded chemical filters.
A Mindful Return to Safe Sun Care
Protecting your skin does not require complicated routines or expensive new tools. It requires intentional, physical touch rather than relying on the quick illusion of a pressurized spray.
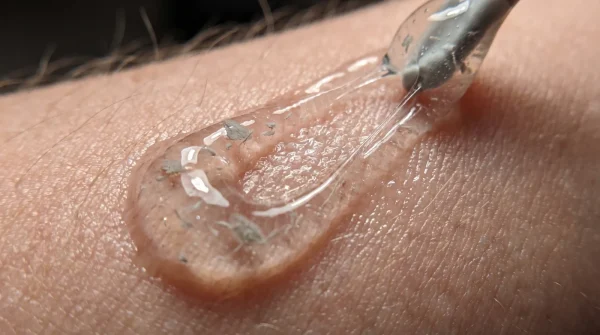
Switching to traditional cream application allows you to monitor the integrity of the product. When you use your hands, you can feel the texture and verify that the product has not separated. The cream should tremble slightly on your fingers, cool and intact, rather than sputtering out like a dying engine.
Creating a new, mindful sun care habit means building a reliable tactical toolkit. Embrace these simple, minimalist actions to guarantee the integrity of your protection against the elements:
- Store all sun care indoors at an ambient temperature of sixty-eight to seventy-two degrees Fahrenheit to prevent early separation.
- Opt for mineral-based creams packaged in squeeze tubes, which rely on stable zinc rather than reactive chemicals.
- Apply your lotion at least fifteen minutes before stepping into the heat, letting it bind safely to your skin without evaporating.
- Use a soft, damp towel to gently wipe away old layers before reapplying, preventing any buildup of oxidized residue.
The True Weight of Intentional Care
Moving away from the quick, hazy mist of an aerosol can feels like giving up a modern convenience. We have grown so accustomed to saving thirty seconds here and there that we forget the quiet satisfaction of properly caring for our bodies through physical touch.
Taking a moment to massage a cool lotion into your arms forces you to slow down. It demands a deliberate pause before you rush out into the summer heat. That small pause, that deliberate choice to avoid the pressurized compromise, brings a profound peace of mind. You are no longer crossing your fingers and hoping for safety; you are guaranteeing it with your own two hands.
The skin does not simply absorb what we give it; it negotiates with every single chemical, and a degraded aerosol leaves your barrier with nothing but a losing battle.
| Key Point | Detail | Added Value for the Reader |
|---|---|---|
| Formula Stability | Creams maintain pH balance longer than pressurized aerosols. | Prevents unexpected chemical burns and skin irritation. |
| Application Control | Manual spreading ensures an even, thick layer of protection. | Eliminates dangerous skipped spots and patchy sunburns. |
| Temperature Resilience | Squeeze tubes do not trap explosive propellants in high heat. | Offers peace of mind when bags are left in the summer sun. |
FAQ
Can I store my spray sunscreen in the fridge?
Extreme cold can also cause chemical separation; room temperature is always the safest choice.
How do I know if my aerosol is degraded?
A sour smell, a watery texture, or immediate stinging upon application are clear warning signs.
Are all spray sunscreens dangerous?
While not all are inherently harmful, the pressurized environment makes any chemical filter highly vulnerable to rapid breakdown.
Does a chemical burn look like a sunburn?
It often appears patchier, stings more sharply, and can develop micro-blisters much faster than typical sun exposure.
What is the best alternative to aerosol sprays?
Mineral-based lotions in standard squeeze tubes provide the most stable, reliable defense against the sun.